Choosing Cytokines for Consistency: A Case Study of FGF-Basic for Use with Pluripotent Stem Cells, from Research to Manufacturing

Cytokines, including growth factors, play an integral role in stem cell research as well as in the development and manufacture of cell therapies. Their role as intercellular and intracellular signaling molecules is critical for expansion and differentiation of a variety of stem and immune cells such as T-cells, NK-cells and iPSC-derived stem cells.
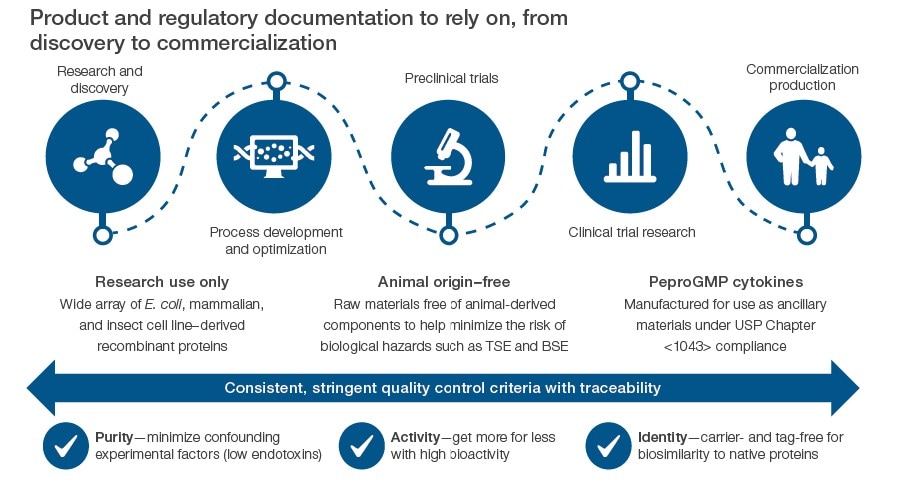
Gibco™ PeproTech™ products include a range of human recombinant proteins to choose from that provide consistent performance including bioactivity and purity. Depending on your lab application, you can select from RUO (research use only) or AOF (animal origin free) for research and development purposes and progress to considering GMP cytokines for preclinical and clinical manufacturing in commercialization of therapeutics.
Case Study of Fibroblast Growth Factor–Basic (FGF-b) for Use with Induced Pluripotent Stem Cells (iPSCs)
Pluripotent stem cells (PSCs) can indefinitely self-renew and retain the potential to differentiate into cells of the three germ lineages: ectoderm, mesoderm, and endoderm [1]. The use of PSCs ranges from basic research to clinical applications and increasingly relies on serum-free and feeder cell–free culture conditions, which depend upon a variety of recombinant proteins. It is crucial that these proteins perform consistently throughout the process, from research to clinical use, to help ensure reliable and reproducible results [2]. Basic fibroblast growth factor (bFGF or FGF-basic), also known as fibroblast growth factor 2 (FGF2), is one of the key recombinant proteins used in stem cell culture. It plays a crucial role in maintaining pluripotency and self-renewal by activating signaling pathways such as PI3K/AKT, PLCγ, and MEK/ERK [3,4].
Ensuring consistent, high-quality bFGF performance is essential for reliable results across stem cell culture workflows, but the lack of clarity regarding the functional equivalence for different bFGF grades poses a challenge for researchers transitioning from research to clinical applications. This study addresses the gap by evaluating three PeproTech bFGF offerings:
- RUO bFGF: Gibco™ Human FGF-Basic (FGF-2/bFGF) (154 aa) Recombinant Protein, PeproTech™ (Product Code: 17851163)
- AOF bFGF: Gibco™ Human FGF-Basic (FGF-2/bFGF) (154 aa) Animal-Free Recombinant Protein, PeproTech™ (Product Code: 17893543)
- PeproGMP bFGF: Gibco™ PeproGMP™ Human FGF-Basic (FGF-2/bFGF) Recombinant Protein, PeproTech™ (Product Code: 30275723)
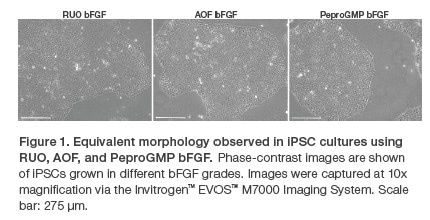
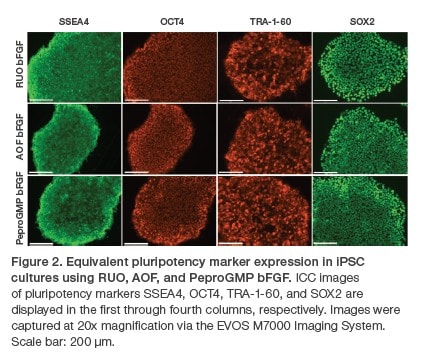
Here, we assess and demonstrate the consistency of the three FGF-b grades by gauging their impacts on the growth and pluripotency of human dermal fiborblast-derived iPSCs. These cells were first expanded over multiple passages in basal medium supplemented with one of the three FGF-b grades, during which growth rate and morphology (figure 1) were observed. Following expansion, their expression of pluripotency markers was measured (figure 2).
The functional equivalence of the three FGF-b grades highlights their ability to integrate across various stages of stem cell research, process development, and clinical manufacturing. This consistent performance from PeproTech human recombinant proteins allows seamless transition between grades without requiring substantial revalidation, helping ensure operational efficiencies while supporting cost-effective workflows (Table 1).
Table 1. Characteristics of the three grades of PeproTech cytokines.
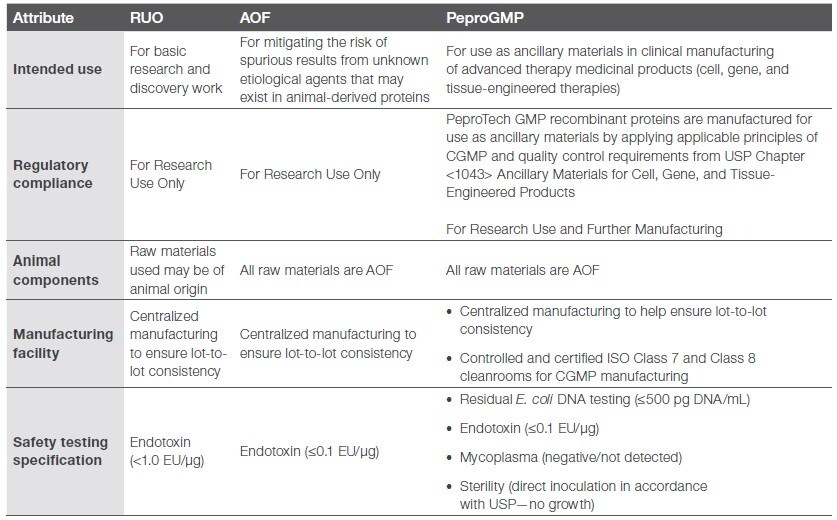
Gibco™ PeproTech™ products can be relied on not only for bFGF grades, but also for other recombinant cytokines and growth factors. PeproTech products are trusted for quality, consistency, and reliability as well as their ability to support the RUO-to-GMP transition. For more information, please visit the PeproTech Product Showcase Page
References
- Melton D (2014) ‘Stemness’: definitions, criteria, and standards. Essentials of Stem Cell Biology. Academic Press. 7–17.
- Kirkeby A, Main H, Carpenter M (2025) Pluripotent stem-cell-derived therapies in clinical trial: a 2025 update. Cell Stem Cell 32(1):10-37.
- Nakashima Y, Omasa T (2016) What kind of signaling maintains pluripotency and viability in human-induced pluripotent stem cells cultured on laminin-511 with serum-free medium? BioResearch Open Access 5(1):84–93.
- Mossahebi-Mohammadi M et al. (2020) FGF signaling pathway: a key regulator of stem cell pluripotency. Front Cell Develop Biol 8:79.