missing translation for 'onlineSavingsMsg'
Learn More
Learn More
Invitrogen™ Pembrolizumab Recombinant Monoclonal Antibody


Description
For reconstitution, add sterile, distilled water to achieve a final antibody concentration of 1 mg/mL. Gently shake to solubilize the protein completely. Do not vortex. Reconstituted products should be stored at -80 °.
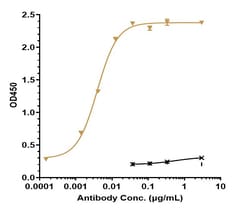
Pembrolizumab (Keytruda) is a humanized monoclonal antibody that is approved by the U.S. Food and Drug Administration for the treatment of patients with unresectable or metastatic melanoma. Pembrolizumab binds to the human cell surface receptor PD-1 (Programmed Cell Death Protein 1) and blocks its interaction with PD-L1, resulting in the activation of T-cell-mediated immune responses against tumor cells.

Specifications
Specifications
| Antigen | Pembrolizumab Humanized |
| Applications | ELISA, Flow Cytometry |
| Classification | Recombinant Monoclonal |
| Concentration | 1 mg/mL |
| Conjugate | Unconjugated |
| Formulation | 25mM histidine with 8% sucrose, 0.01% Tween 80 and no preservative; pH 6.2 |
| Gene Alias | Keytruda; MK-3475 |
| Host Species | Human |
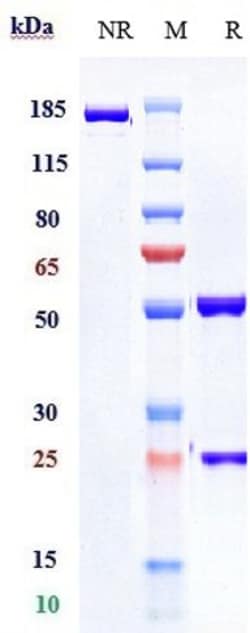
| Purification Method | Protein A |
| Quantity | 100 μg |
| Show More |
Product Title
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.
Spot an opportunity for improvement?