missing translation for 'onlineSavingsMsg'
Learn More
Learn More
Invitrogen™ Ranibizumab Humanized Recombinant Human Monoclonal Antibody


Description
Endotoxin level < 0.01EU/μg by LAL method Use a manual defrost freezer and avoid repeated freeze thaw cycles. Store at 2 to 8 °C for one week. Store at -20 to -80 °C for twelve months from the date of receipt.
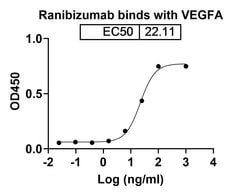
Ranibizumab with trade name Lucentis, is an FDA-approved drug for the treatment of patients with neovascular (Wet) age-related macular degeneration (AMD) and macular edema following retinal vein occlusion (RVO). It is a recombinant humanized IgG1 kappa isotype monoclonal antibody fragment. Ranibizumab binds to vascular endothelial growth factor A (VEGF-A) and blocks its activity.

Specifications
Specifications
| Antigen | Ranibizumab Humanized |
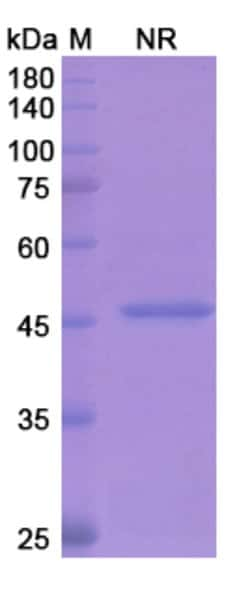
| Applications | ELISA, SDS-Page |
| Classification | Recombinant Monoclonal |
| Concentration | 1.5 mg/mL |
| Conjugate | Unconjugated |
| Formulation | PBS with no preservative; pH 7.4 |
| Gene Alias | Fab-12 variant Y0317; Lucentis; RhuFab |
| Host Species | Human |
| Immunogen | Human VEGFA. |
| Purification Method | Protein L |
| Show More |
Product Title
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.
Spot an opportunity for improvement?